
Journal Information
 Malaria 2000
Malaria 2000
 Issue 10
Issue 10
 Previous Issues
Previous Issues
 Instructions to Authors
Instructions to Authors
 Staff
Staff
 Your comments
Your comments
Other Journal Resources
 Malaria and African links
Malaria and African links
 Congress
Congress
 Submit an event
Submit an event
 News
News
Journal Quick Search
Issues under development
 Forum
Forum
 African Malaria Society
African Malaria Society

K.M. Bhatt, S.M. Bhatt, A.J. Oloo, S.A. Jivanjee, C. Kanja, B. Kimanzi and W.S. Ekissa
A study on efficacy and tolerance of Mefloquine was carried out in three large centres in Kenya, the Kenyatta National Hospital in Nairobi, Coast General Hospital in Mombasa and New Nyanza Hospital in Kisumu. A total of 257 patients with uncomplicated acute falciparum malaria were treated with 1000 mg of mefloquine in two split doses 8 hours apart. 253 patients completed the study. All had negative blood clearance time was 18 hours and the mean parasite clearance time was 48 hours. 81 patients from Nairobi were followed up until day 28 and all were cured. The patients in Kisumu. and Mombasa were not followed up until day 28 as these are highly endemic areas and it would have been impossible to rule out reinfection. The drug was well tolerated, the main side effects being dizziness which was noted in 74 patients.

Malaria continues to be an increasingly important problem particularly in Africa. In tropical Africa about 370 million people live in endemic areas where Plasmodium falciparum is the dominant parasite. Chloroquine is the most commonly used first line drug against malaria in the African continent. However spread of chloroquine resistance has been reported in most areas in Africa South of Sahara, including West Africa (1,2,3). In Rwanda for instance in 1985 only 31% of patients showed some grade of resistance to chloroquine whilst in 1987 the resistance had doubled to 65% (2). Varying degree of chloroquine resistance in Kenya has been reported especially along the coastal region and in Western Kenya (4-7). Currently the chloroquine resistance in some areas may be between 50 - 75% (8-10). The increase in resistance to chloroquine has lead to change in recommendation in prophylaxis for tourists visiting Africa. Currently the most commonly used prophylactic antimalarial in Africa and other countries, where chloroquine resistant falciparum malaria exists, is mefloquine. Mefloquine is also widely used for the treatment of acute malaria where multidrug resistant falciparum malaria occurs and even high dose mefloquine is used in some areas. (11-13).
In South East Asia artemesinin compounds are increasingly used due to emergence of resistance to other existing antimalarials. However high recrudescence rate was noted when artemisinin compounds were used alone but the cure rate improved remarkably when mefloquine was added to the therapy. (14,15).
Mefloquine has been used by tourists visiting Kenya for a long time. In view of the very high incidence of malaria in Kenya and rapid spread of resistance to most of the commonly available antimalarials it is critically important that newer and hopefully more effective drugs be tested. Mefloquine is one such drug which has already been studied in some parts of East Africa (16-19) and has recently been introduced in Kenya market.
Mefloquine - like several other antimalarial drugs, mefloquine is a distance derivative of quinine. The common feature of all these drugs is the bicycle conjugated quinoline right system. The mechanism of action of mefloquine is not yet fully understood. It is known that mefloquine binds with high affinity to erythrocyte membrane and may thus possibly block the invasion of uninfected erythrocytes by merozoites. It appears that the action of mefloquine is linked to the breakdown of hemoglobin by the Plasmodia, leading to their destruction by lethal membrane damage. The mode of action of quinine, chloroquine and mefloquine are very similar in many respects but unlike the first two compounds, mefloquine does not intercalate into the DNA. Mefloquine is a blood schizontocidal drug for Plasmodium falciparum malaria. It also exerts a gametocytocidal effect on Plasmodium malariae, P ovale and P. vivax but not on the mature gametocytes of P. falciparum. There is no effect on exoerythrocytic hepatic forms and sporozoites, Mefloquine has a long elimination half life of 20 days.
The study was an open clinical trial using the drug mefloquine (Mephaquin Lactabs supplied by Mepha Ltd., Aesch-Basel Switzerland). The study was carried out in three large centres in Kenya, the Kenyatta National Hospital in Nairobi, Coast General Hospital in Mombasa and New Nyanza Hospital in Kisumu. The patients were recruited into the study if they were diagnosed as having acute, uncomplicated falciparum malaria, were above the age of twelve years and when a consent was obtained to take part in the study. Reasons for exclusion were pregnancy, severe and complicated malaria (20), history of antimalarial treatment within preceding ten days, mixed infections, children under 12 years and presence of other obvious infections.
All patients were admitted to hospital for five days and thereafter were followed up as out patients on days 7,14, 21 and 28. A complete physical examination was carried out on each patient on admission and body temperature, pulse and respiratory rates were recorded every 6 hrs. Signs and symptoms were evaluated every day for the first five days and weekly thereafter. The main signs and symptoms evaluated were fever, headache, joint pains and myalgia.
Pretreatment investigations included full blood count, blood sugar, scrum electrolytes, scrum bilirubin, alkaline phosphatase, blood urea, creatinine, albumin globulin, aspartate and alanine amino-transferase and urine analysis. These tests were repeated on days 14 and 28. Parasitological examination of thick and thin blood film was done every 24 hrs for the first 5 days and thereafter on days 7,14 and 28. Only the patients from Nairobi were followed up until day 28. Mombasa and Kisumu patients were followed up until day 14 as both these areas are highly endemic for malaria and reinfection cannot be ruled out in long term follow up.
Fever clearance was taken as the time from the start of treatment until axillary temperature dropped to 37.5°C and remained below this temperature during the next 48 hours. Blood films were considered negative if no parasites were seen in 200 oil immersion fields in a thick blood film.
Patients were treated with 1000 mg of mefloquine in two split doses 8 hrs apart. Side effects were defined as signs and symptoms that first occurred or became more severe after treatment was started. Cure was defined as the absence of recrudescence during 28 days of follow up.
A total of 257 patients were studied between the three centres. 100 in Nairobi, 85 in Mombasa and 72 in Kisumu. 253 patients completed the study. 4 patients defaulted from the study before completing seven days. There were nine other patients who were excluded from the study. Six patients in Nairobi had mixed infection with P. vivax and P. falciparum and one patient in Nairobi and two patients in Mombasa had pure P. vivax infection. All were Ethiopians. The mean age of the study population was 25.3 years, youngest being 12 years and oldest 70 years. There were 162 males and 95 females. The mean parasite count prior to treatment was 1.8 x 104 per µl. The distribution of parasite density is as indicated in Fig 1.
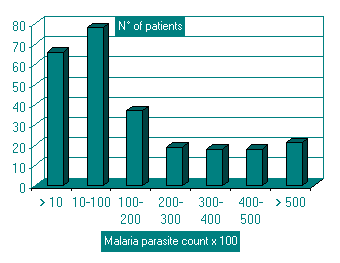
Fig 1: Distribution of Malaria parasite count
The mean fever clearance time was 18 hrs and parasite clearance time was 48 hrs. Fig 2 shows the Mean parasite counts after treatment.
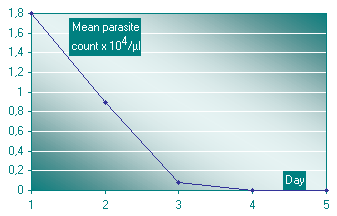
Fig 2: Mean parasite counts
Two patients took longer than three days to clear parasites. Both these patients were found to be HIV positive. There were no significance derangements in hematological parameters serum creatinine, blood sugar, urea electrolytes, or any of the liver function tests before or after treatment (Table I). Three patients had slightly raised alanine aminotransferase levels on day 7 and two on day 14 but all had normalised by day 28. All the patients completed follow up upto day 7 and had negative slides.
|
MEAN (SD) |
ADMISSION MEAN (SD) |
DAY 14 MEAN (SD) |
DAY 28 MEAN (SD) |
|
Packed cell volume % |
34 (4) |
33 (3) |
36,7 (5,4) |
|
White Blood cell count (cells/µl) |
8186 (2926) |
8183 (3927) |
7295 (1057) |
|
Platelets |
310 x 103 |
274 x 103 |
227 x 103 |
|
ALAT |
44,9 (30,2) |
54.1 (20,6) |
42,0 (11) |
|
Serum creatinine |
106 (7) |
112 (14) |
109 (11) |
|
Total Protein |
77 (12) |
79 (10) |
76 (14) |
|
Albumin |
37 (7) |
36 (4) |
36 (4) |
|
Blood Glucose |
5,7 (1,2) |
|
|
Table I: Labaratory parameters
81 patients all, from Nairobi completed follow up until day 28. All these patients were cured (Table II).
|
FOLLOW UP |
N° OF |
POSITIVE |
NEGATIVE |
|
Day 7 |
253 |
0 |
253 |
|
Day 14 |
167 |
0 |
167 |
|
Day 21 |
97 |
0 |
97 |
|
Day 28 |
81 |
0 |
81 |
Table II: Follow up of patients and their blood slides
The commonest adverse effect of the drug was a feeling of dizziness observed in 74 patients which occurred within the first two days of starting treatment and lasted 2-3 days. The other effects observed were as per table III.
|
|
Number |
Starting day |
Duration in days |
|
Dizziness |
74 |
0-1 |
2-3 |
|
Headache |
25 |
3-4 |
1-2 |
|
Abdominal pain |
12 |
0-1 |
1-3 |
|
Nausea/vomiting |
5 |
1-2 |
1 |
|
Diarrhoea |
3 |
2 |
1-2 |
|
Itching |
3 |
2 |
2-3 |
|
Loss os appetite |
3 |
1 |
6 |
Table III: Episodes of adverse reactions
This study was carried out on patients representing a wide spectrum of clinical disease and varying immune status, Nairobi being a non endemic area for malaria, majority of the normal residents are non immune and acquire infection from elsewhere, whereas both Mombasa and Kisumu. are endemic areas of malaria and the residents have some immunity. The additional observation in Nairobi was a group of refugees from Ethiopia and Somalia with borderline nutrition status among many of them. However as far as the efficacy of mefloquine is concerned there was no difference noted in the response rate in any of the centres studied. There were two patients who took longer than three days to clear their parasites, who subsequently were found to be HIV positive. No definite conclusion can be drawn from this observation as we did not study the HIV status of all the patients. The study showed a 100% cure upto day seven, unfortunately not all the patients completed the follow up upto day 28 but those who did complete the study the cure rate was 100%.
Although we confined out treatment to the protocol, in some instances we did use mefloquine in very sick patients on humanitarian grounds due to lack of other drugs, the response to treatment in these very sick patients was excellent. The drug was fairly well tolerated by most patients, dizziness being the main side effect which lasted 2-3 days with complete recovery. As all our patients were in hospital in bed, the dizziness did not bother them much and a large number of the patients did not volunteer the information unless they were specifically asked. However for out patient treatment it is advisable to warn the patients of this possible side effect as it may be disturbing in our study population. The rare neuropsychiatric side effects observed in some instances have been related to much higher doses used. In this study we have used a lower dose and split the dose further into two doses 8 hours apart.
Since the majority of our patients treated with mefloquine were uncomplicated malaria with normal biochemical parameters, it was an opportunity to study any derangements of these parameters on treatment with mefloquine. There were no significant changes noted in any of the major organ functions.
1000 mg of mefloquine given in 2 split doses 8 hours apart was found to be very effective in treatment of falciparum malaria and fairly well tolerated. However the drug should be appropriately used for correct indication. Mefloquine is also a very useful drug for prophylaxis particularly for short term (2). It is an ideal drug to use in patients who are on short term artemisinin compounds to prevent recrudescence (22). Indiscriminate use of mefloquine may encourage development of resistance as it has a very long half life and therefore it should be one of the second line treatments for malaria. Any observations on emerging resistance should be reported immediately.
-
Watkins, W. M, Oloo, A. J, Mjomba, M, Koech, D.K. and Gilles, H.M. Chloroquine resistant falciparum malaria responsiveness to treatment with Halofantrine. Lancet 1988, 11; 251.
-
Garcia-Vidal Joan, Ngirabega, J., Soldevila, M., Navarro R and Bada L. Evaluation of resistance of Plasmodium falciparum to antimalarial drugs in Rwanda, 1985 - 1987. Trans. Roy. Soc. Trop. Med. Hyg., 1989, 83:490.
-
Lepers, J. P., Deloron, P., Mouden, J.C. Bras, J and Coulanges, P. Sudden. Increase in number of isolates of Plasmodium falciparum resistant to chloroquine. In Madagascar. Trans. Roy. Soc. Trop. Med. Hyg., 1989, 83:491-492.
-
Masaba S.C. and Spencer H.C. Sensitivity of Plasmodium falciparum to chloroquine in Busia District Trans. Roy. Soc. Trop. Med. Hyg., 1982 76: 314.
-
Spencer H. C, Masaba S. C. and Kiaraho D. Sensitivity of Plasmodium falciparum isolates to chloroquine in Kisumu and Malindi Kenya. Amer. J. trop. Med. Hyg., 1982 31: 902.
-
Oloo AJ, Owanga M, Kamuvi F, Watkins WM and Spencer HC. Susceptibility to chloroquine in Kisumu area Western Kenya. Proc. of 5th Annual Medical Scientific Conference KEMRI/KETRI 1984 Pap No 68/84.
-
Watkins WM, Spencer HC. Sixsmith Dg and Koech DK. Susceptibility of Plasmodium falciparum with Chloroquine and Amodiaquine at Malindi and Msa Msambweni Kenya Coast. Abst. Proc. of 5th Ann Med. Sci Conf. KEMRI/KETRI 1984 Abstract No. 132.
-
Spencer, HC., Kipingor T, Aguere R., Koech DK and Chulay JD. Plasmodium falciparum in Kisumu Kenya: Differences in sensitivity to Amodiaquine and chloroquine in vitro. Journal of infection diseases, 1983 148: 732-736.
-
Oloo, AJ, Adongo A., Kariuki D., Boriga DA, Magirl C, Were JB and Koech DK. Sensitivity of falciparum malaria to chloroquine and amodiaquine in four districts of Western Kenya (1985-1987). EAMJ 1991 606-610.
-
Keuter M, Sanders J, Kamotug H., Ronday M, Schoufer E, Vetkamps S, Khalurni G, Ngwana W., Wetsleyn JCFM., de Gens A., Bradly - Bennett AD. Parasitological, Clinical and hematological response of children with Plasmodium falciparum to 4-aminoquinolines and to pyrimethamine -sulfadoxine with quinine in Western Kenya. Trop. Geog. Med., 1992 44:1-8.
-
Harinasuta T, Bummag D, Wernsdorter, WH. A Phase 11 clinical trial of mefloquine in patients with chloroquine resistant falciparum malaria in Thailand. Bull WHO, 1983 6: 299-305.
-
Kofi Ekwe M. A double blind comparative clinical trial of mefloquine and chloroquine in symptomatic falciparum malaria. Bull WHO, 1983 61: 713-718.
-
WHO advances in malaria chemotherapy. Technical report series 711 Geneva WHO, 1984.
-
Looarcesuwan S, Viravan S, Vanijanonta S, Wilairatona P, Suntharasamal P, Charoenlarp P, Arnold K, Kyle DE, Canfield C, Webster K,A randomised trial of mefloquine, artesunate and artesunate followed by mefloquine in acute uncomplicated falciparum malaria. Lancet, 1992 1: 821-824.
-
Looareesuwan S, Kyle DE, Virvan C, Vanijanonta S, Wilairatana P, Charoenlarp P, Canfield CJ, Webster HK. Treatment of patients with recrudescent falciparum malaria with a sequential combination of artesunate and mefloquine Am. J. Trop. Med. Hyg., 1992, 47: 794-799.
-
Wasame M, Lebbod M, Ali S, Wensdorfer WH and Bjorkman A. Susceptibility of Plasmodium falciparum to chloroquine and Mephaquine in Somalia. Trans. Roy. Soc. Trop. Med. Hyg., 1988 82: 202-204.
-
Kilimani V.A.E.B., Mkufya AR and Kilama WL. Low resistance of Plasmodium falciparum to Mefloquine in Tanga region, Tanzania. Trans. Roy Soc. Trop. Med. Hyg., 1989 83; 162-164.
-
Warsame M, Wensorfer WH, Payne D and Bjorkman susceptibility of Plasmodium falciparum in vitro to chloroquine, mefloquine, quinine and sulfadoxine/Pyrimethamine responses to the different drugs. Trans. Roy. Soc. Trop. Med. Hyg., 1991, 85; 565-569.
-
Warsame M. Wernsdorder, WH, Willcox W., Kulane AA and Bjorkman. A. The changing pattern of Plasmodium falciparum susceptibility to chloroquine but not to mefloquine in a mesoendemic area of Somalia. Trans. Roy. Soc. Trop. Med., 1991, 85; 200-203.
-
Warrell DA, Molyneux ME, Beales PF, severe and complicated malaria 2nd edition. Trans. Roy. Soc. Trop. Med. Hyg., 1990 84: (SuppI 2) 1-65.
-
WHO International Travel and Health 1995.
-
Looareesuwan S, Wilcuratnap, Vanijanota S and Viravan C. Treatment of acute uncomplicated falciparum malaria with a short course of artesunate followed by mefloquine. South East Asian Journal of tropical medicine and public health 1993, 24: 230-234.
| PDF file |
Home | Top of Page | Malaria 2000 | Previous Issue | Links | Congress | Contact Us